Home Acid Base CO2 in Ocean Electronegetivity Electron affinity Ionic charge Ionisation energy Molecular geometry Polyatomic ions Practical Ptable Std Re Potential Titration
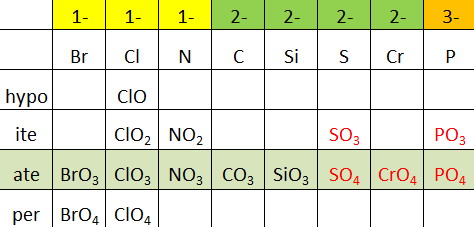
| 1- | 2- | 3- | 1+ | 2+ |
| acetate, CH 3 COO – | carbonate, CO 3 2- | arsenate, AsO 3 3- | ammonium, NH 4 + | dimercury, Hg 2 2+ |
| bromate, BrO 3 – | chromate, CrO 4 2- | phosphite, PO 3 3- | hydronium, H 3O+ | |
| chlorate, ClO 3 – | dichromate, Cr 2 O 7 2- | phosphate, PO 4 3- | ||
| chlorite, ClO 2 – | hydrogen phosphate, HPO 4 2- | |||
| cyanide, CN – | oxalate, C 2 O 4 2- | |||
| dihydrogen phosphate, H 2 PO 4 – | peroxide, O 2 2- | |||
| hydrogen carbonate, HCO 3 – | silicate, SiO 3 2- | |||
| hydrogen sulfate, HSO 4 – | sulfate, SO 4 2- | |||
| hydrogen sulfide, HS – | sulfite, SO 3 2- | |||
| hydroxide, OH – | ||||
| hypochlorite, ClO – | ||||
| nitrate, NO 3 – | ||||
| nitrite, NO 2 – | ||||
| perchlorate, ClO 4 – | ||||
| permanganate, MnO 4 – |